Key Takeaways:
- RHP integrates exposure science, toxicology, and epidemiology to evaluate potential human health risks from chemical exposures.
- Our scientists develop defensible scientific analyses that support regulatory decisions, product stewardship, and litigation.
- Multidisciplinary expertise allows RHP to evaluate human health hazards, quantify exposures, and characterize potential risk across the full product lifecycle.
- Clients rely on RHP for objective, scientifically rigorous insights that withstand regulatory and legal scrutiny.
Integrated Science to Evaluate Chemical Risks and Support Regulatory, Legal, and Corporate Decision-Making
RHP supports clients across industry, government, and legal sectors by translating complex scientific evidence into clear insights that withstand scrutiny from regulators, courts, and scientific peers.
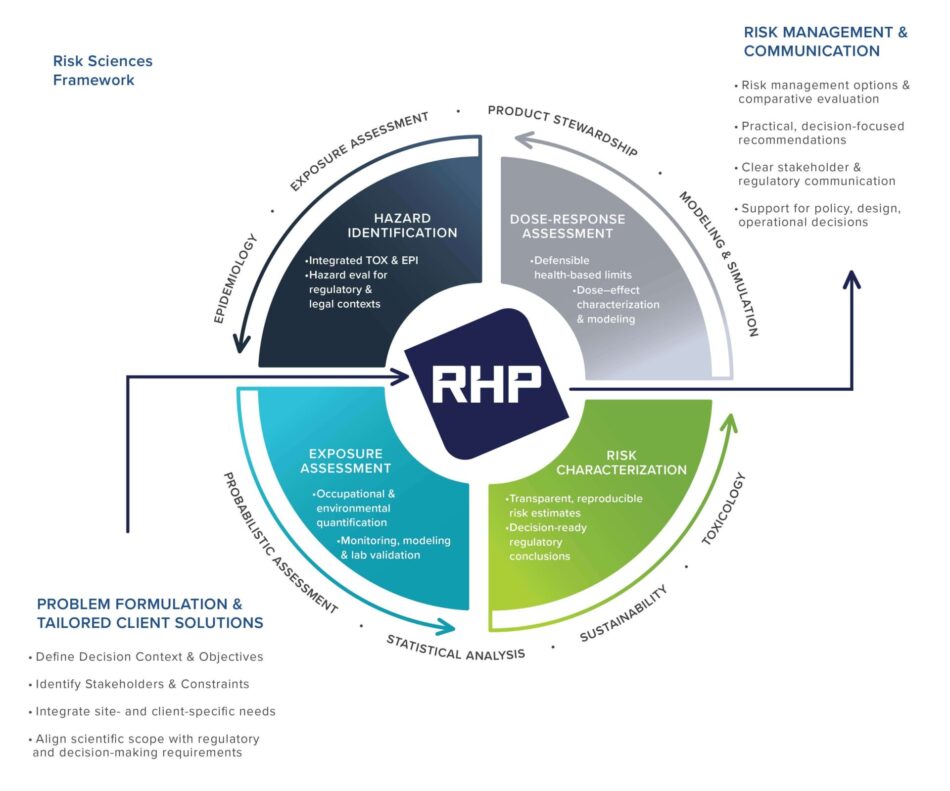
Organizations evaluate potential human health risks from chemical exposures across manufacturing, consumer product use, and environmental pathways. Addressing these challenges requires defensible science that integrates exposure data, toxicological evidence, and epidemiologic research.
RHP’s Human Health Risk Sciences (HHRS) practice combines expertise in exposure science, toxicology, epidemiology, and risk assessment to evaluate hazards, quantify exposures, and characterize potential human health risk.
Our multidisciplinary teams apply scientifically accepted methods to develop reliable data and evidence-based conclusions. Some of these methods include:
- Exposure modeling;
- Environmental sampling;
- Laboratory testing;
- Dose-response analysis;
- Systematic review and hazard characterization;
- Benchmark dose modeling;
- Physiologically based pharmacokinetic (PBPK) modeling; and
- Risk characterization and communication.
Integrated Human Health Risk Science
Understanding potential human health risks requires evaluating multiple lines of scientific evidence. RHP integrates three core disciplines to provide comprehensive assessments of potential human health risks.
RHP scientists measure and model real-world chemical exposures in occupational, environmental, and consumer settings. Using laboratory testing, monitoring, and exposure modeling, we quantify exposures and reconstruct historical scenarios to support risk assessments and regulatory decisions.
Our scientists evaluate toxicological and epidemiologic evidence to determine whether chemical exposures can cause adverse health effects and to assess the strength of exposure-disease relationships. These analyses support hazard identification, dose-response evaluation, and causation assessments used in regulatory and legal contexts.
RHP supports product safety and regulatory compliance by evaluating chemical risks throughout the product lifecycle. Our scientists assess exposure potential, toxicological hazards, and regulatory requirements to help organizations develop safer products and manage emerging chemical risks.
How RHP’s Human Health Risk Sciences Supports Clients
- Integrated evaluations of potential human health risks across exposure science, toxicology, and epidemiology.
- Assessment of chemical hazards and exposure pathways across occupational, environmental, and consumer settings.
- Development of defensible exposure and risk data to support regulatory decisions and product stewardship.
- Scientific analyses and expert support for regulatory, litigation, and public health decisions.
Contact RHP experts to reduce uncertainty and enable confidence in decision-making by delivering reliable, reproducible, and relevant data. Through early and active stakeholder engagement, we provide customized solutions that support clients with their specific goals. Call RHP at (866) 481-8188.
