Key Takeaways:
- RHP integrates toxicological and epidemiologic evidence to evaluate chemical hazards.
- Our scientists apply dose-response analysis and weight-of-evidence frameworks to characterize potential human health risks.
- RHP experts examine the totality of scientific literature to assess evidence of causation for specific human health outcomes.
- We perform critical reviews of scientific and regulatory evidence to support defensible conclusions for regulatory and legal decisions.
- RHP translates complex scientific findings into clear, decision-ready insights for clients.
Authoritative Human Health Risk Science to Interpret Evidence, Assess Causation, and Support Regulatory, Legal, and Risk Management Decisions
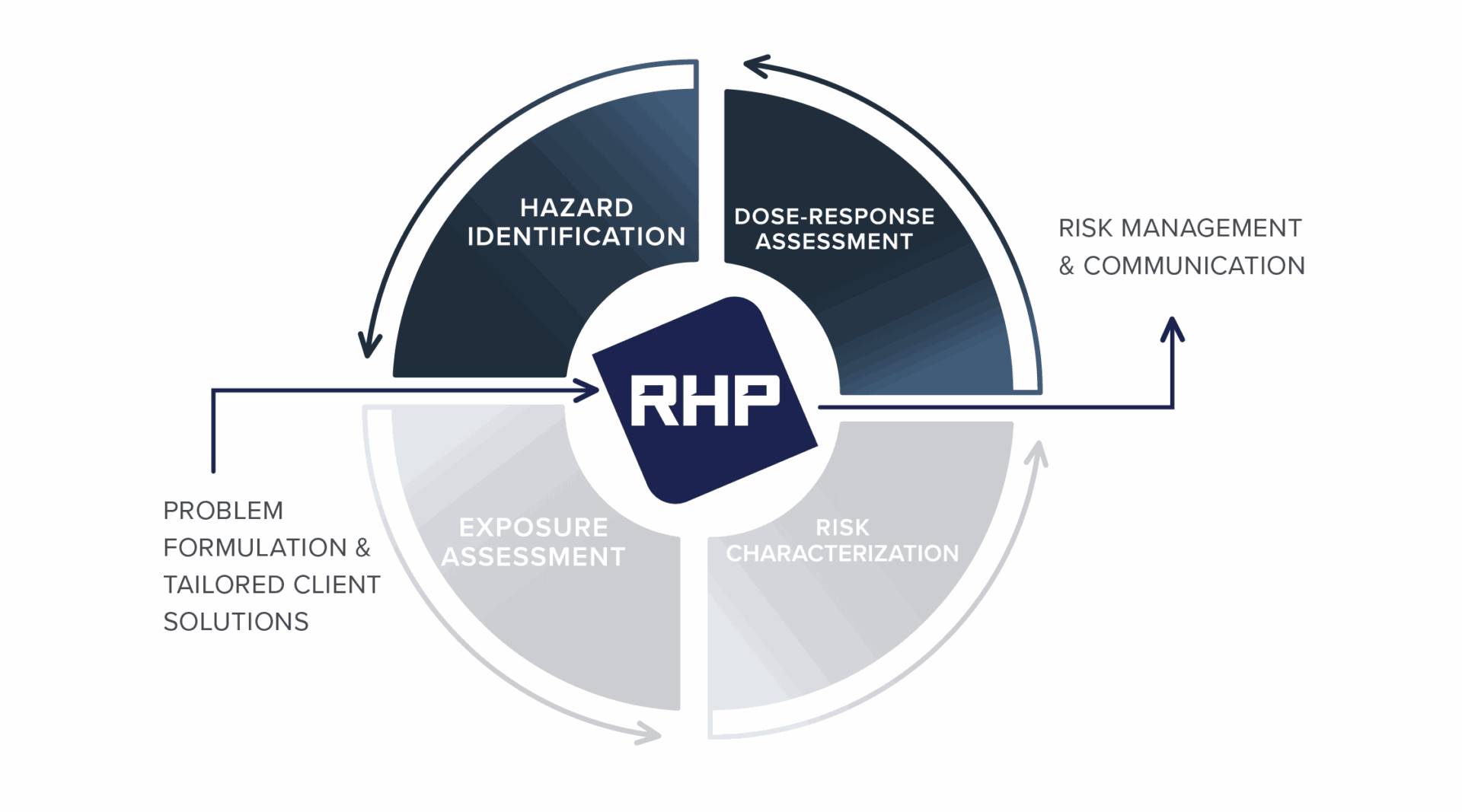
Understanding human health risks from chemical exposures requires integration of toxicological evidence, epidemiologic data, and exposure science. RHP Risk Management combines expertise across these disciplines to evaluate complex scientific questions and translate evidence into clear, defensible conclusions.
Our scientists analyze toxicological and epidemiologic evidence to assess chemical hazards, dose-response relationships, potential human health risks, and causation across occupational, environmental, and consumer exposure scenarios. These analyses support regulatory compliance, product safety and stewardship strategies, public health decision-making, and litigation.
By applying scientifically accepted methods – including dose-response evaluation, benchmark dose modeling, new approach methodologies (NAMs), and systematic review of scientific literature – RHP delivers objective, evidence-based insights that withstand scrutiny from regulators, courts, and scientific peers.
By applying scientifically accepted methods – including dose-response evaluation, benchmark dose modeling, new approach methodologies (NAMs), and systematic review of scientific literature – RHP delivers objective, evidence-based insights that withstand scrutiny from regulators, courts, and scientific peers.
Integrated Approach to Human Health Risk Science
RHP toxicologists evaluate laboratory and mechanistic evidence to determine whether chemical exposures can cause adverse health effects and to characterize the relationship between dose and response.
Our epidemiologists analyze population-based data to evaluate associations between environmental exposures and health outcomes, integrating complex datasets to develop consensus-based interpretations of the scientific evidence.
By integrating toxicology, epidemiology, and exposure science, RHP develops comprehensive human health risk assessments that inform regulatory strategies, product safety and stewardship decisions, and litigation support.
What RHP Delivers
- Integrated evaluation of toxicology and epidemiology evidence.
- Critical review of scientific and regulatory literature.
- Dose-response and population-level risk assessment.
- Evaluation and causation and weight-of-evidence frameworks.
- Scientific support for regulatory, legal, and corporate decision-making.
Contact RHP experts to reduce uncertainty and enable confidence in decision-making by delivering reliable, reproducible, and relevant data. Through early and active stakeholder engagement, we provide customized solutions that support clients with their specific goals.

